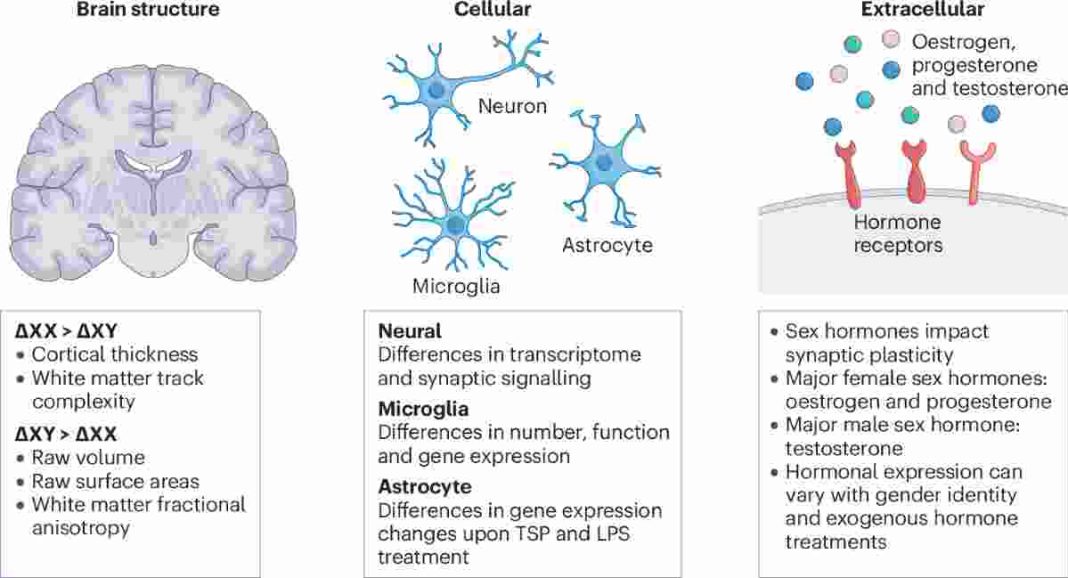
A groundbreaking study has uncovered widespread differences in how genes function inside individual brain cells of men and women, offering fresh insights into why certain neurological and psychiatric disorders affect the sexes differently. Published in the journal Science on April 16, 2026, the research analyzed gene expression at single-cell resolution across more than a million brain cells from 30 adults — 15 men and 15 women — providing the most detailed map yet of sex-based molecular variations in the human cerebral cortex.
Led by neurogeneticist Alex DeCasien at the National Institute on Aging, the team examined 169 postmortem brain samples from six cortical regions known to show subtle volume differences between sexes on neuroimaging scans. Using advanced single-nucleus RNA sequencing, researchers found that while men and women have remarkably similar numbers and proportions of neurons and glial cells, the activity levels of thousands of genes inside those cells vary significantly by sex. More than 3,000 genes displayed sex-biased expression in at least one brain region, with 133 genes showing consistent differences across multiple areas and cell types.
The strongest variations appeared in genes located on the X and Y sex chromosomes, as expected. However, the majority of differences involved autosomal genes not tied to sex chromosomes, likely influenced by sex steroid hormones such as estrogen and testosterone. These patterns were most pronounced in the fusiform cortex, as well as in oligodendrocytes, astrocytes, and excitatory neurons. Even though biological sex accounted for less than 1% of the overall variation in gene expression, the targeted differences were strategically located in pathways linked to brain function and disease vulnerability.
Many of the genes showing sex-biased activity overlap with genetic risk factors for conditions that disproportionately affect one sex. These include ADHD and schizophrenia, which tend to show higher prevalence or different presentations in males, and Alzheimer’s disease and depression, which often impact females more severely. The findings suggest that these molecular distinctions at the cellular level could help explain observed sex differences in disease risk, progression, and symptoms, moving beyond broad brain structure observations toward precise cellular mechanisms.
Importantly, the study does not claim that male and female brains are entirely different or that one is superior. Instead, it highlights subtle, region-specific, and cell-type-specific tweaks in gene regulation that accumulate across the cortex. Experts describe the dataset as a “treasure trove” for future research, enabling scientists to investigate whether these variations arise purely from genetics and hormones or also reflect lifelong effects of socialization and experience. Future prenatal or early developmental studies could help disentangle these influences.
The research builds on earlier bulk-tissue analyses but provides far greater resolution by zooming in on individual cells. Traditional brain imaging or whole-tissue studies often mask these fine-grained differences, which single-cell techniques now bring into sharp focus. No major differences emerged in overall cell counts or basic brain architecture between the groups, reinforcing that sex effects operate more at the level of molecular orchestration than gross anatomy.
This new atlas of sex effects on brain gene expression arrives at a pivotal time for personalized medicine. By identifying specific genes and cell types where male and female brains diverge, the work opens pathways to develop sex-tailored treatments for neurological disorders. Pharmaceutical developers could use the data to design interventions that account for hormonal influences or chromosome-linked mechanisms, potentially improving outcomes for patients of both sexes.
The study also underscores the value of including balanced representation of male and female samples in neuroscience research, a practice that has gained momentum but still requires broader adoption. As scientists continue to mine this rich dataset, they expect to uncover additional layers of how biological sex shapes brain function at the most fundamental level.
While the differences remain relatively modest overall, their consistency and strategic placement suggest they play meaningful roles in everyday brain operations and long-term health. The publication has already sparked excitement among researchers studying neurodevelopmental, psychiatric, and neurodegenerative conditions, who see it as a foundational resource for understanding and eventually addressing sex disparities in brain health.
As the scientific community digests these findings, the focus shifts toward translating this molecular knowledge into clinical advances. From more precise diagnostic tools to hormone-informed therapies, the implications could reshape how medicine approaches brain disorders in the years ahead. This detailed view of gene activity in male and female brain cells marks a significant step toward a more nuanced, biologically informed understanding of human brain diversity.